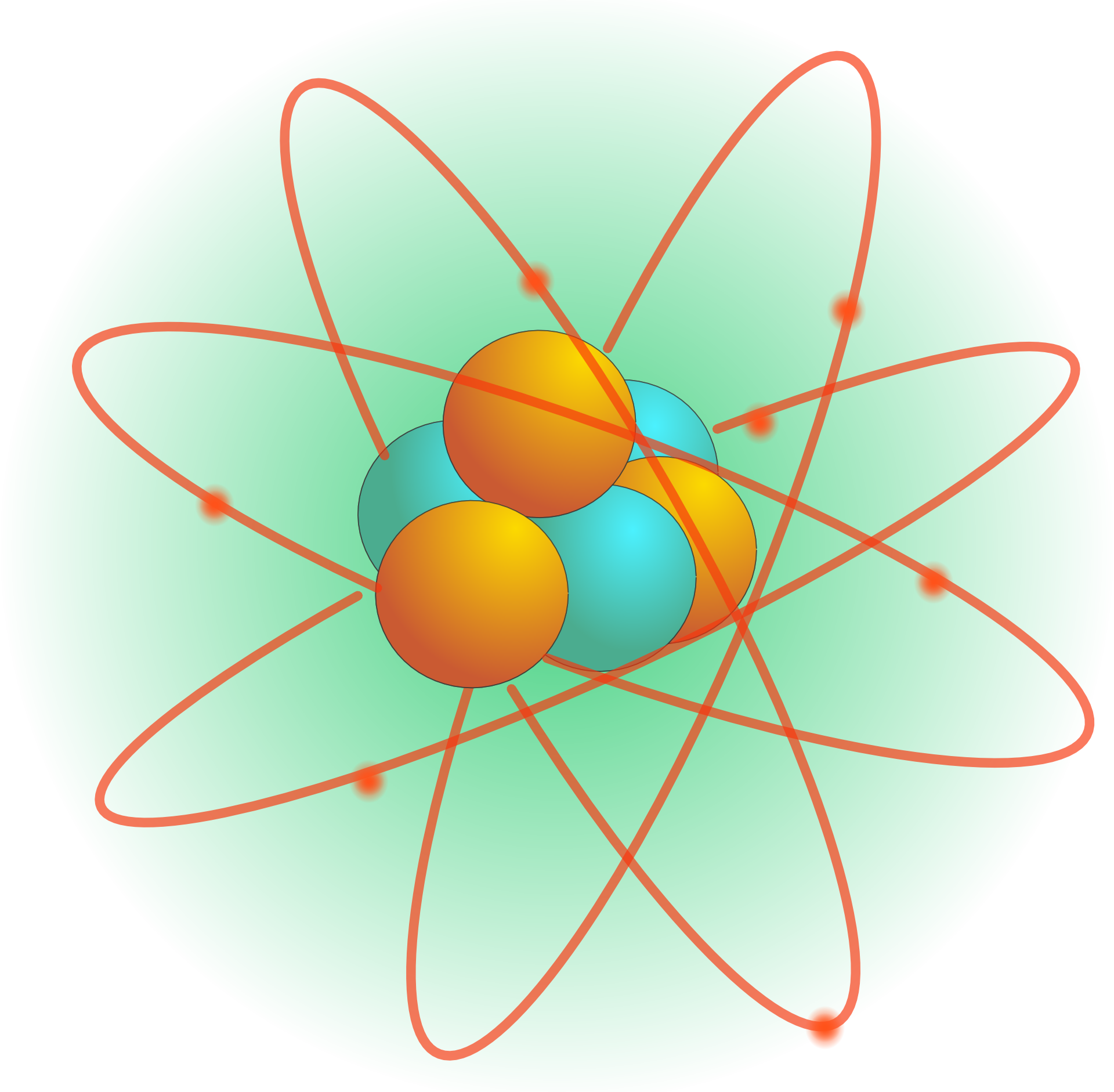 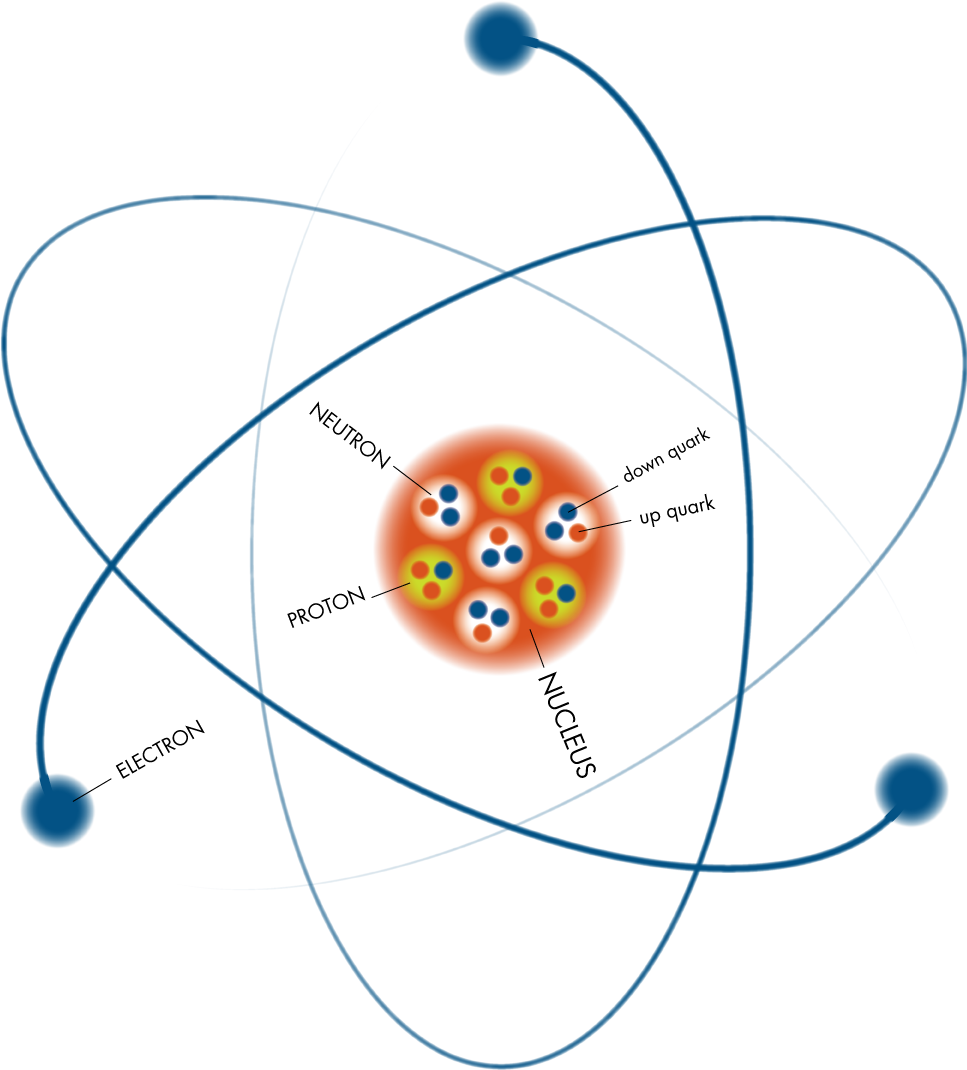 
Atoms have different properties based on the arrangement and number of their basic particles. The outermost regions of the atom are called electron shells and contain the electrons (negatively charged). The nucleus (center) of the atom contains the protons (positively charged) and the neutrons (no charge). Atomic ParticlesĪtoms consist of three basic particles: protons, electrons, and neutrons. Many biological processes are devoted to reassembling molecules into different, more useful molecules. For example, water is composed of hydrogen and oxygen atoms that have combined to form water molecules. Atoms combine to form molecules, which then interact to form solids, gases, or liquids. It is equal in mass to a proton or it weighs 1 amu.Īn atom is the smallest unit of matter that retains all of the chemical properties of an element. neutron: A subatomic particle forming part of the nucleus of an atom.proton: Positively charged subatomic particle forming part of the nucleus of an atom and determining the atomic number of an element.atom: The smallest possible amount of matter which still retains its identity as a chemical element, consisting of a nucleus surrounded by electrons.Neutrons are uncharged particles found within the nucleus.Each electron has a negative charge (-1) equal to the positive charge of a proton (+1).Protons and neutrons have approximately the same mass, about 1.67 × 10 -24 grams, which scientists define as one atomic mass unit (amu) or one Dalton.An atom is composed of two regions: the nucleus, which is in the center of the atom and contains protons and neutrons, and the outer region of the atom, which holds its electrons in orbit around the nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
